
Enabling a paradigm shift in medicine and research Imagine a future where drugs are developed without the use of animals and where personalised replacement organs are designed and built, rather than harvested from donors. Aspect Biosystems is working to make this vision a reality. We are an award-winning Canadian biotechnology company operating on the leading edge of 3D bioprinting and tissue engineering with our patented microfluidic bioprinting platform technology. We place our RX1™ bioprinters in strategic academic research labs around the world to enable the development of tissues for regenerative medicine, disease modeling and drug discovery applications. We also actively collaborate with industry partners to co-develop tissues for drug discovery and implantation.
A unique tissue fabrication technology based on microfluidics
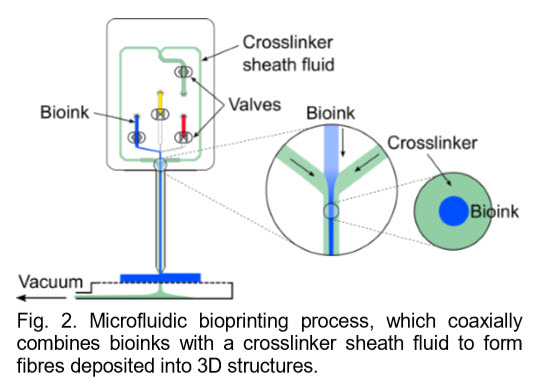
We have taken concepts in 3D printing, lab-on-a-chip, computer-aided-design and tissue engineering to realize the most advanced 3D bioprinting technology to date. Our RX1™ platform powered by patented microfluidic technology, can programmatically manipulate various bioinks combined with living human cells and then deposit those living materials layer-bylayer in geometrically intricate ways. This enables rapid printing of macro-scale 3D structures that incorporate intricate micro-level details to generate architecturally and functionally accurate human tissues.
Our microfluidic bioprinting platform has the following unique advantages:
• Rapid switching and combining of biomaterials: Microfluidic technology enables the rapid switching between different biomaterial inputs in real time to rapidly build heterogeneous tissues that recreate physiological structures without having to change printheads.
• Cell phenotype is preserved and viability is high: Unlike the high-shear environments present in inkjet or extrusion bioprinting devices, our microfluidic printing generates cell-loaded fibres with very low shear stress due to the shielding effect from coaxial flow focusing. Fragile primary human cells are printed at high viabilities and human iPSCs can be printed without negative cell responses such as cell death or premature differentiation.
• Proprietary microfluidic printheads for printing vasculature: With our core-shell printheads, cell-laden perfusable hollow fibres with three different shell layers can be directly printed within larger 3D printed tissues. This effectively enables users to pattern blood vessel-like networks within larger tissues, which is essential in maintaining cell viability and function.
• Flexible biomaterial options: The microfluidic technology underpinning the RX1™ is highly flexible with respect to biomaterial choices. Printable biomaterials include various natural and synthetic hydrogels and tissue-specific bioinks from decellularized matrices to provide a highly physiologically-relevant microenvironment for the cells. Printing of rationally-designed biomaterials such as modified multi-arm PEG-based materials is also enabled.
• Portfolio of plug-and-play printheads: The first generation Aspect printheads support 1-4 bioinks + 1 crosslinker + 1 buffer solution. We are developing a portfolio of advanced plug-and-play printheads compatible with the RX1™ platform with increased material options and novel functions, enabling users to easily upgrade functionality without having to modify the bioprinter.
• Partnership focused approach: We work with our partners in a unique way; academic collaborators form important nodes in a Discovery Ecosystem of users who are provided with high levels of expert support to ensure their tissue printing programs are successful, evidenced by journal publications.
Functional tissue applications Our contractile airway muscle tissue is the first and only 3D printed human airway smooth muscle tissue that contracts rapidly and appropriately in response to physiological stimuli, including histamine, and relaxes in response to bronchodilator drugs, such as salbutamol, in vitro. These responses are highly comparable to those seen in ex vivo airway tissues and airways in vivo. This contractile tissue platform is also being expanded to other tissues including intestinal, vascular and skeletal muscle. The rapid printing, precise and reproducible cell positioning in 3D, high cell viability and preserved phenotype, and the ability to customize bioink compositions, make Aspect’s RX1™ bioprinting platform highly applicable to the biofabrication of ground-breaking 3D tissues for research and clinical applications:
Fig. (a) A printed, contractile ring with human airway smooth muscle cells (HASMs), (b) HASMs in printed fibres (live/dead, day 16) and (c) contraction and relaxation in response to physiological doses of histamine and salbutamol.
1. iPSC-derived neural tissues: Printed human iPSCs maintain viability and expression of pluripotency markers post-printing. iPSC-derived neural progenitor cells express higher levels of neural markers post-printing compared to extrusionbased bioprinting.
2. Perfusable tissues: Cell-laden, multishell hollow perfusable fibres can be printed for the biofabrication of a physiological, perfusable tissues, such as liver tissues for predicting drug-induced liver injury.
3. 3D pancreas: ESC-derived pancreatic clusters can be printed into living 3D structures with potential use as a therapeutic implant for patients with diabetes.
4. 3D tumor models: Cancer cells can be printed with healthy cells to study cell interactions in vitro and to provide 3D tumor models for drug screening. The developed glioblastoma tumor model exhibits upregulated levels of cancer stem cell and metastatic invasiveness markers compared to 2D cultures.
5. 3D meniscus: A printed knee meniscus tissue exhibits the appropriate biomechanical properties for implantation.






